/ff336b12-ba9a-433f-b65c-320c61a28fe5.png)
Clinical Trial Budget Template - Simplify Study Budgeting with Subtotal and Investigator
Review Rating Score
Planning a clinical trial and need to manage your budget effectively? Look no further! At BizzLibrary.com, we are here to assist you with our comprehensive Clinical Trial Budget Template. This template is designed to help you track and manage the financial aspects of your clinical trial, ensuring that you stay on track and make informed decisions.
The Importance of a Clinical Trial Budget
A well-planned budget is crucial for the success of any clinical trial. It allows you to allocate resources efficiently, plan for expenses, and ensure that your study remains financially viable. Here are some key reasons why a clinical trial budget is essential:
- Accurate Financial Planning: A budget helps you anticipate the expenses associated with the clinical trial, from the costs of study materials and equipment to participant compensation and travel expenses. By estimating and organizing these costs, you can create a realistic financial plan.
- Resource Allocation: With a budget in place, you can allocate your resources effectively, ensuring that each aspect of your trial is adequately funded. This includes budgeting for personnel, research materials, laboratory services, and any other factors necessary for the study's success.
- Identifying Potential Funding Opportunities: A comprehensive budget provides a clear overview of the total costs involved in your clinical trial. This allows you to identify potential funding sources, such as grants, sponsorship, or collaborations, to cover the expenses and ensure the continuation of your research.
- Financial Compliance: By maintaining a detailed budget, you can adhere to financial regulations and guidelines set by regulatory bodies and funding agencies. This ensures transparency and accountability, minimizing the risk of financial mismanagement or non-compliance.
Key Features of our Clinical Trial Budget Template
Our Clinical Trial Budget Template offers a user-friendly and comprehensive solution for managing your trial's finances. Here are some key features:
- Study Details: Capture important study information such as the study title, start and end dates, and a brief description.
- Time Allocation: Plan and track the allocation of your study budget over time, taking into account the duration of each phase or milestone.
- Fee Breakdown: Break down the anticipated fees for various aspects of the study, such as site monitoring, data management, ethics submissions, and statistical analysis.
- Subtotal Calculation: Easily calculate subtotals for each budget category, allowing you to monitor the overall expenditure for different aspects of your trial.
- Investigator Payments: Include a section to track payments to investigators, ensuring accurate and timely compensation.
Download our Clinical Trial Budget Template
Take control of your clinical trial finances and set yourself up for success. Download our Clinical Trial Budget Template in DOCX format today. It's a valuable tool for any clinical research professional looking to manage their trial's budget efficiently.
Visit BizzLibrary.com now to explore a wide range of business document templates, including budget templates, study agreements, and more. Simplify your clinical trial management and achieve the financial organization your study deserves!
Is the template content above helpful?
Thanks for letting us know!
Reviews
Lynwood Andrews(8/3/2023) - DEU
Grateful!!
Last modified
Our Latest Blog
- The Importance of Vehicle Inspections in Rent-to-Own Car Agreements
- Setting Up Your E-mail Marketing for Your Business: The Blueprint to Skyrocketing Engagement and Sales
- The Power of Document Templates: Enhancing Efficiency and Streamlining Workflows
- Writing a Great Resume: Tips from a Professional Resume Writer
Template Tags
Need help?
We are standing by to assist you. Please keep in mind we are not licensed attorneys and cannot address any legal related questions.
-
Chat
Online - Email
Send a message
You May Also Like

Corporate Development Agenda Template - Streamline and Enhance Development Initiatives Globally

Letter Of Transmittal Template - Create a Professional Project Transmittal Letter
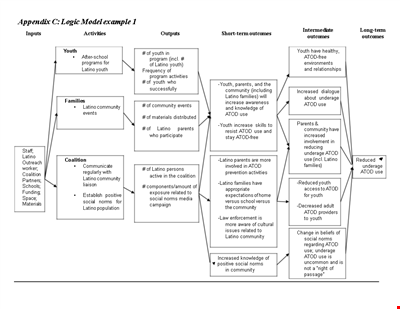
Logic Model Template for Community Youth Latino

Sales Proposal Template: Create Winning Presentations for Clients

Creating a Personal Birth Plan Template for a Happy and Following Birth with Your Partner

Meeting Proposal Templates - Video-Focused | JordannaRajaProposal.com

Creating an Effective Informational Interview Agenda for a Successful Career Meeting

Client Website Design Proposal Agreement - Professional Website Design Services

Sample Proposal Collection: Explore Project Options for Inspiration

Lawn Care Services Proposal for Landscaping

Project Business Proposal Template | Word | Streamline Your Project Business

Nonprofit Program Budget Template | Project Budget for Organization

Simple Marketing Strategy Template for Effective Planning and Execution

College Website Design Proposal - Creative and Functional Website Solutions

Construction Engineering Work Order Template | Easy Project Management

Free Sales Action Plan Example PDF Template for Download